Rubidium creates a few compounds with other chemical elements, but it is used rather for research than in industry. Name: Rubidium Symbol: Rb Atomic number: 37 Atomic weight: 85.468 State: solid Group, period, block: 1, 5, s Color: grey white Classification: alkali metal Electron configuration. The atomic radii of first group elements are given below : Group-I element Atomic Radii (pm) Na 86. (ii) Of these four elements, sodium, lithium and rubidium are all in Group 1. In nature, rubidium is found in some minerals like leucite, carnalite, and others, and it is among 25 most common chemical elements on the surface of our planet. Rubidium was discovered in 1861 by German chemists Robert Bunsen (1811-99) and Gustav Kirchhoff (1824-87), in the mineral lepidolite through the use of a spectroscope. Down a Group: Atomic radius increases down any Periodic Table Group because. Its properties in living organisms are similar to the ones of potassium. 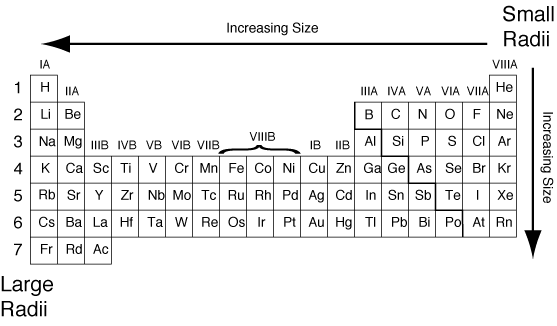
This element is non-toxic and it has no biological importance but it can be found in our body is really very small amounts. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius. 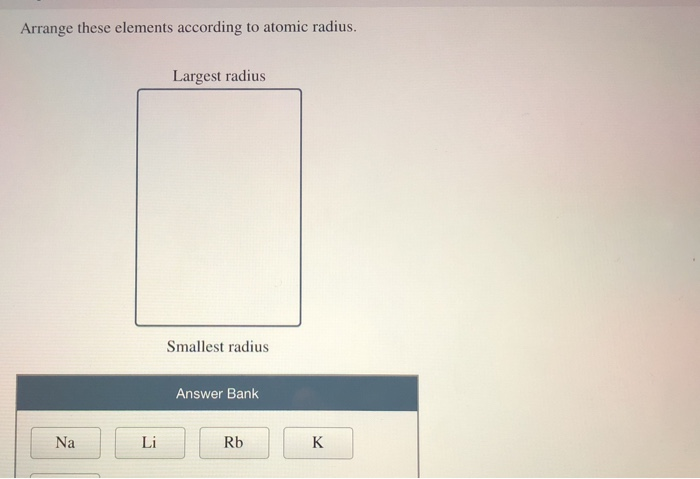
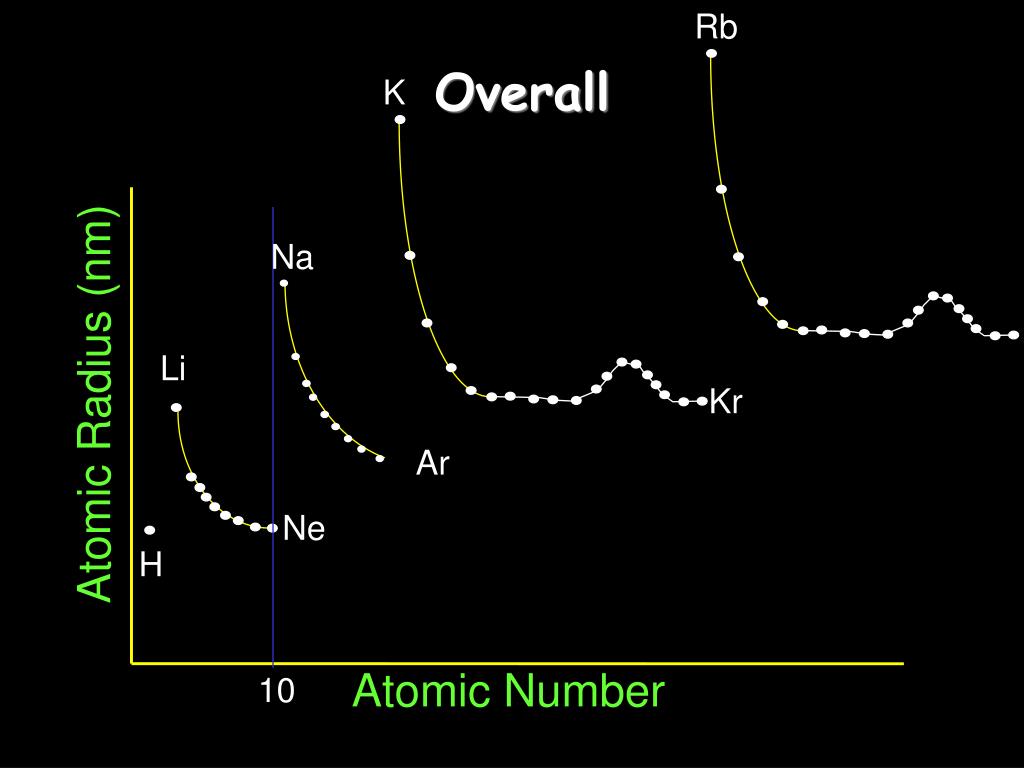
atoms in order of increasing first ionization energy: F, Li, N, Rb. The atomic radius of a chemical element is a measure of the size of its atom, usually the mean or typical distance from the center of the nucleus to the outermost isolated electron. Its name is derived from a Latin word meaning a deep red color. Within each period, the trend in atomic radius decreases as Z increases for example. Rubidium is a soft and quite reactive metal. This problem has been solved In the following graphs, pick the one that best predicts the atomic radius of Rb 296 243 200 190 Atomic radii (pm) Li Na K 190.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
